|
8/16/2023 0 Comments Serial dilution 
Medical and pharmaceutical personnel are constantly dealing with dosages that require concentration measurements and dilutions. 
So each mix has a dilution-factor times less than the one before it. 1:2 in this example) so the concentration (relative amount of the thing to the non-thing) decreases exponentially. Of course, the addition of the stock solution affects the total volume of the diluted solution, but the final concentration is likely close enough even for medical purposes. The objective of the serial dilution method is to estimate the concentration (number of colonies, organisms, bacteria, or viruses) of an unknown sample by counting the number of colonies cultured from serial dilutions of the sample, and then back track the measured counts to the unknown concentration. How do serial dilutions work In a serial dilution you perform a series of stepwise dilutions where you’re diluting by the same factor each time (e.g. Carefully pipet up and down twice to mix. Pipet 40 L of the original blue dye into the first well (C1). The sample/culture is taken in a test tube and six test tubes, each with 9 ml of sterile diluents, which can either be distilled water or 0.9 saline, are taken. Preparing Five-Fold Serial Dilution (Dilution Factor of Five) Using a new pipet tip, pipet 160 L of DI-water into the first 5 wells of row C (C1-C5). 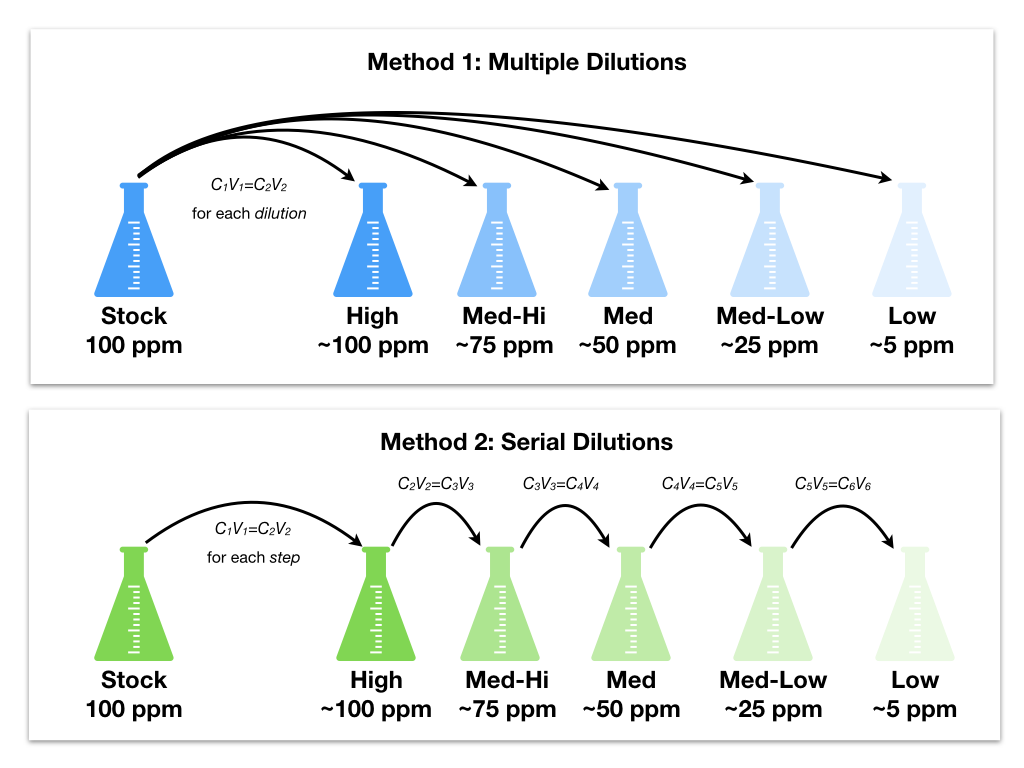
If the stock solution is 10.0% KCl and the final volume and concentration need to be 100 mL and 0.50%, respectively, then it is easy to calculate how much stock solution to use: The following is the procedure for a ten-fold dilution of a sample to a dilution factor of 10-6. Solution Exercise 15.4 Example of Using Standard Line Equation to Determine CFU of a Sample Solution Exercise 15.5 Exercise 15. Medical personnel commonly must perform dilutions for IV solutions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
